- Home
- About Us
- Work
- Blog
- Journal
- Contact
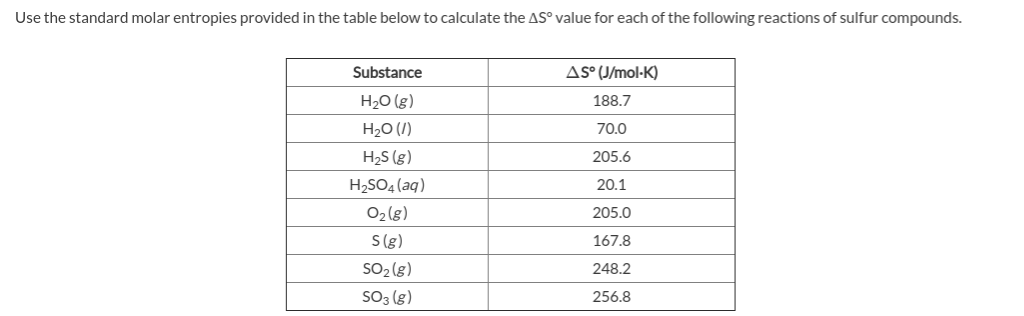
- Standard molar entropy
- Barn finds forza horizon xbox 360
- Star next to someone on xbox friends list
- Vlf receiver lightning detector
- Dmx albums full
- Awara bengali movie download
- Delphi 7 enterprise full
- Sims 4 cc cat ears and tail
- Code of va search warrants
- 3-5 character sheet fillable
- Sl loney trigonometry solutions pdf
- Vray light material
- Charlie murder count smockula brain
- Google sketchup gratis en espa-ol
- Python autoclicker
- Home
- About Us
- Work
- Blog
- Journal
- Contact
- Standard molar entropy
- Barn finds forza horizon xbox 360
- Star next to someone on xbox friends list
- Vlf receiver lightning detector
- Dmx albums full
- Awara bengali movie download
- Delphi 7 enterprise full
- Sims 4 cc cat ears and tail
- Code of va search warrants
- 3-5 character sheet fillable
- Sl loney trigonometry solutions pdf
- Vray light material
- Charlie murder count smockula brain
- Google sketchup gratis en espa-ol
- Python autoclicker

Standard partial molar free enthalpies, enthalpies, and entropies of vaporization from infinitely dilute solutions in Apiezon M were calculated from retention volumes determined over a range of temperatures data are listed for many organic and a few MR4 compounds (M = Si, Ge, and Sn), including Ge(C2H5)4.

The design of a flow system for the measurement of the heat capacity of solutions (Picker et al., 1971) has provided valuable information on aqueous solutions. A similar identity between X3 and X3 (x3 - 0) occurs for the volumes and heat capacities but not for the chemical potentials and entropies. Calorimetric data have the advantage that, as theory shows, the standard partial molar enthalpy H3 for a solute in solution is equal to the partial molar enthalpy in the infinitely dilute solution, i.e. However, disagreement between published A//-functions for apolar solutes in aqueous solutions may also stem from practical problems associated with low solubilities (Gill et al., 1975). Standard chemical potential jj°, standard partial molar enthalpy h°, and standard partial molar entropy s,° for a few hydrated ions Standard state 101.3 kPa, 298 K, unit activity in molality scale.Ĭalculation of A//e -quantities from the dependence of AG on temperature is less reliable than direct calorimetric measurements (Franks and Reid, 1973 Frank, 1973 Reid et al., 1969). Table 9.3 shows the numerical values of the standard chemical potential, the standard partial molar enthalpy h°, and the standard partial molar entropy. The standard chemical potentials of various hydrated ions tabulated in electrochemical handbooks are thus relative to the standard chemical potential of hydrogen ions at unit activity in aqueous solutions. of hydrogen ions is set zero as the level of reference for the chemical potentials of all other hydrated ions. In electrochemistry we make it a rule that the standard chemical potential ju. The standard partial molar Gibbs free energy of solution is related to the enthalpy and entropy functions at the column temperature T by the expression. Note that thermodynamic tabulations do not normally report the standard partial molar properties of solutes and G p, but rather the enthalpy of formation. The only real solution along the Henry s-law line is the inhnitely dilute solution, so that. pp.Thus, the partial molar enthalpy along the Henry s-law line is constant and equal to the standard partial molar enthalpy.
Chloroethane Main page (Russian) Search in database (English) Sitemap (Russian) Properties of substance: chloroethane Group of substances:organic
